Bursa size, an easy tool to evaluate gumboro vaccination programmes
11 February 2022
By conducting necropsies routinely, we can have a rough but quick idea of what is happening on our farms. However, most of the time we do not standardise or record the results of those necropsies. Recording the bursa size as a routine practice can be used to evaluate the Gumboro situation on farms and in particular the vaccination scheme we are using.
The bursa of Fabricius (BF) is a primary lymphoid organ that plays a key role in the differentiation of B-lymphocytes and thus the development of the humoral immunity. Its development begins during incubation and reaches maximum size between 8 and 10 weeks of age, when the regression process starts, and is completed by 6 to 7 months of age (Olah et al., 2014).
The immature B-lymphocytes are the target cells for replication of the IBDV (Infectious Bursal Disease Virus, also known as the Gumboro disease virus).
This is why the bursa of Fabricius is the principal diagnostic organ for evaluation of the Gumboro situation on farms.
When the IBDV replicates in the lymphocytes, we will normally see an impact on the bursa size. This impact can range from an increased size because of oedema (at the beginning of the replication; Figure 1a), to a decrease in size or atrophy due to the disappearance of most of the lymphocytes in the bursa (at the end of replication; Figure 1b).
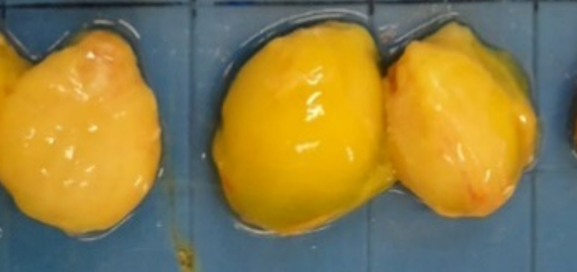 Figure 1. a) Impact on the bursa size due to the replication of IBDV: Oedematous bursas (increased size).
Figure 1. a) Impact on the bursa size due to the replication of IBDV: Oedematous bursas (increased size).
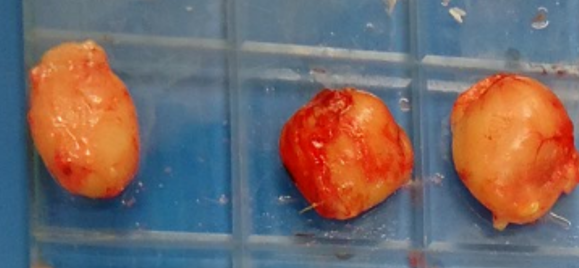 Figure 1. b) Atrophic bursas (decreased size).
Figure 1. b) Atrophic bursas (decreased size).
Accordingly, live IBD vaccines will also have an impact on the bursa size as an effect of vaccine virus replication. In fact, this vaccine replication is one of the most important protective mechanisms of live IBD vaccines against the disease, thanks to the competitive exclusion principle. However, in this case, the impact on the bursa size is less marked, with only a certain decrease in size normally observed at the end of the replication.
Building up an historical record of the bursa size in all our flocks can be very helpful in detecting the time when the bursas start to decrease in size as a consequence of the vaccine replication and its correlation with the MDAs. In this way, if we detect a delay in some flocks (or even that there is no atrophy at all), we can speculate that we are facing a problem or that the MDAs were initially higher than usual (Table 1).
It is important to remember here that when using an HVT-IBDV recombinant vaccine, we do not expect to observe any impact on the bursa size (as there is no live attenuated IBDV; Table 1). So, when these vaccination programmes are used, any change in the bursa size might be indicative of a field Gumboro virus replication.
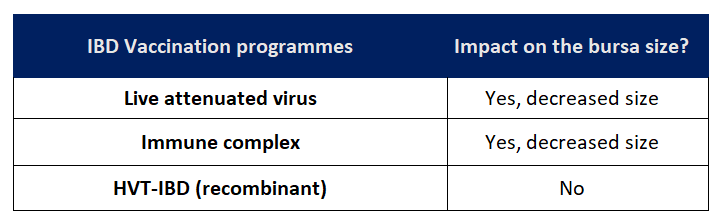 Table 1. Impact on bursa size of different IBD vaccination programmes.
Table 1. Impact on bursa size of different IBD vaccination programmes.
There are several methods of evaluation of the bursa size. We can score the size of the bursa using a bursa-meter (Figure 2) or we can simply measure the bursa with a ruler. When using a bursa-meter, we will obtain a score from 1 to 8 depending on the diameter of the bursa (Figure 2).
Scores from 4 to 6 are considered normal for a healthy bursa (including live attenuated IBDV replication). Scores below or above that (1, 2 and 8) may be indicative of a pathological condition of the bursa.
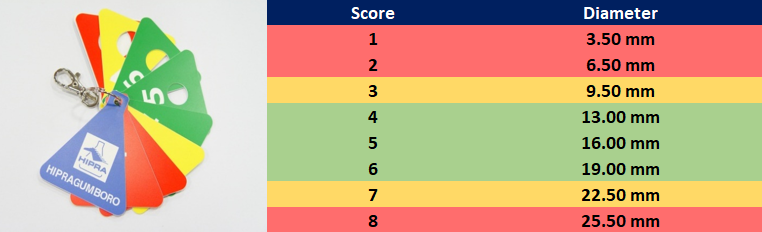 Figure 2. Images and scale of HIPRA bursa-meter.
Figure 2. Images and scale of HIPRA bursa-meter.
As has been explained, the size of the bursa can be a good indicator of the time when the bursa started to be atrophic as a consequence of the vaccine replication. However, it is important to take into account that, as a lymphoid organ, there are other causes (infectious and non-infectious conditions) that can also affect bursa size (Table 2).
For this reason, a differential diagnosis will be crucial when the size and the time of the atrophy observed on the farm does not correlate with what is expected after vaccination.
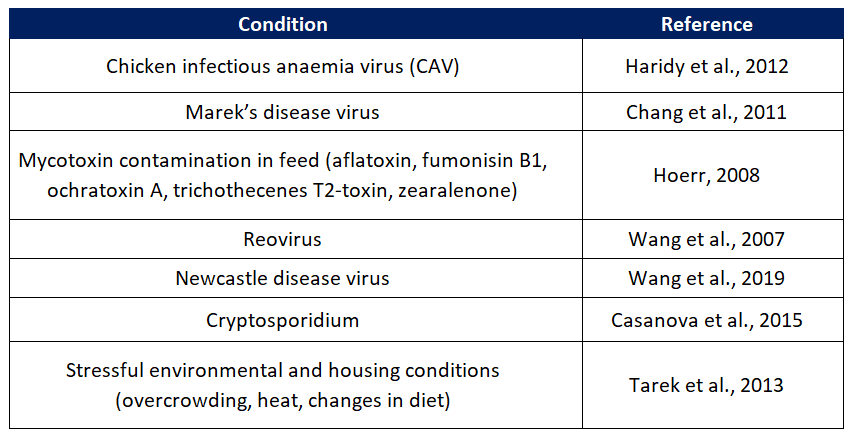 Table 2. Review of different conditions that can directly impact the size of the bursa of Fabricius.
Table 2. Review of different conditions that can directly impact the size of the bursa of Fabricius.
In addition, before using the bursa of Fabricius size as an indicator, it is also important to set size standards for a healthy bursa of Fabricius. Unfortunately, there are not many up-to-date publications that include all types of breeds, genders or farming conditions (Table 3).
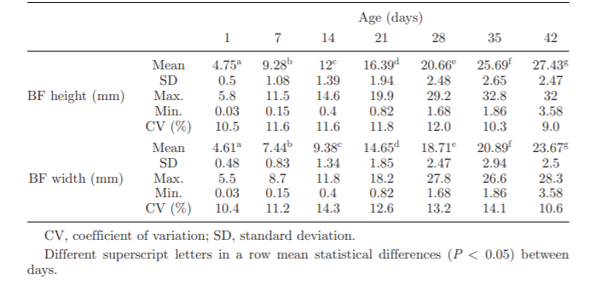 Table 3. Bursa of Fabricius height (mm) and width (mm) in male Cobb 500 broilers (Cazaban et al; 2015).
Table 3. Bursa of Fabricius height (mm) and width (mm) in male Cobb 500 broilers (Cazaban et al; 2015).
This is why it is so important for a historical record to be built up for each farm, as this parameter will probably just be comparable within this same unit.
Don't miss any updates
Controller: LABORATORIOS HIPRA, S.A.
Purposes: Managing the contractual and/or business relationship with HIPRA, including sending news, promotions and invitations to events sponsored by HIPRA.
Lawful basis: Performance of the contractual relationship and HIPRA’s legitimate Interest.
Recipients: Third parties to which HIPRA has entrusted cloud computing, security, auditing, mailing, technical and computer support services, as well as companies in its group.
Rights: Request access to and rectification or erasure of personal data and other rights as explained in the additional information. You can seeview the detailed additional information about data protection in our Privacy Policy.
For further information, please check our detailed information on Data Protection.